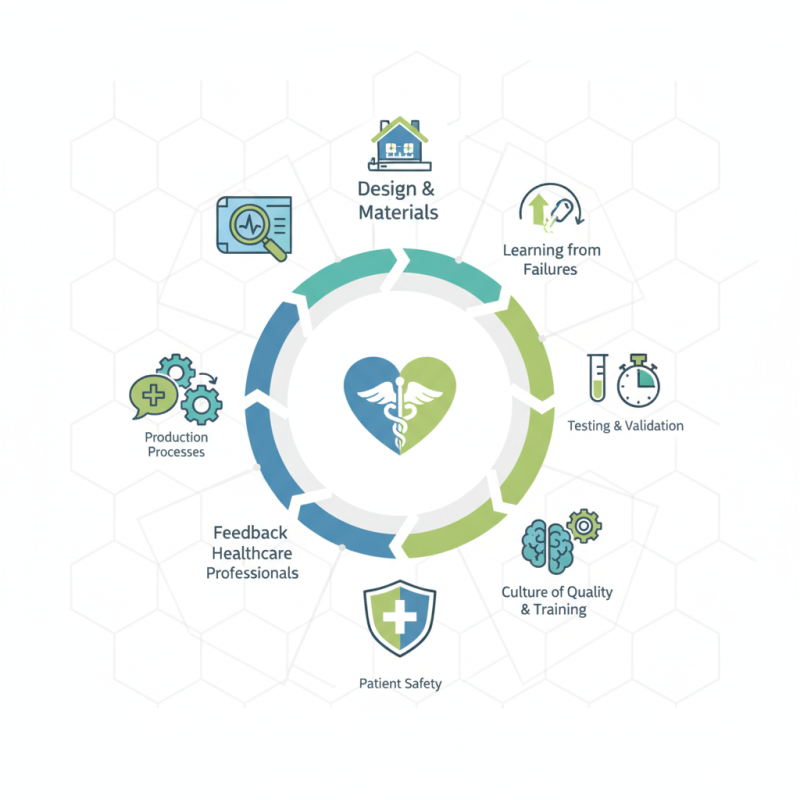
Ensuring key factors in medical device quality is crucial for patient safety and effective treatments. The medical device industry is complex, involving multiple stakeholders. Manufacturers must prioritize quality in every stage of product development. This means understanding the intricacies of design, materials, and production processes.
A strong focus on key factors in medical device quality can help mitigate risks. Quality control measures should be implemented rigorously. Testing and validation processes must be thorough. However, failures can happen. Learning from past mistakes is vital for improvement. Companies must reflect on their practices regularly.
Building a culture of quality within an organization is essential. Training and continuous education for employees can enhance awareness. Feedback from healthcare professionals can provide valuable insights. Balancing innovation with quality assurance presents challenges. Striving for excellence while acknowledging limitations is necessary for growth in this field.
In the medical device industry, quality standards are crucial. The International Organization for Standardization (ISO) provides extensive guidelines. ISO 13485 outlines requirements for a quality management system, ensuring consistent design and manufacturing processes. Adhering to this standard can prevent significant risks in product performance.
Data shows that up to 70% of recalls stem from quality-related issues. This highlights the need for rigorous testing and validation protocols. Companies must establish robust documentation practices to track compliance. Additionally, risk management strategies, as specified by ISO 14971, can streamline the identification of potential hazards associated with medical devices. This proactive approach can mitigate risks effectively.
Ignoring these standards can lead to devastating consequences. The FDA reports that nearly 1 in 5 devices face quality issues post-market launch. It calls for continuous improvement and transparency in the quality management process. Organizations should invest in training staff on these standards. This fosters a culture committed to excellence in quality and safety, which is non-negotiable in the medical device sector.
In the medical device industry, implementing effective quality management systems (QMS) is crucial. A robust QMS can enhance product safety and efficacy, ensuring compliance with regulatory standards. According to a report by the FDA, 70% of medical device recalls are linked to quality issues. This statistic underlines the importance of diligent quality control throughout the production process.
Developing a QMS requires a detailed approach. Begin with defining quality objectives. Ensure these align with industry standards, such as ISO 13485, which provides a framework for quality management specific to medical devices. Conducting regular audits and updates of the QMS is essential. This keeps the system relevant and effective.
**Tip:** Establish a cross-functional team dedicated to quality oversight. Diverse perspectives will help identify potential gaps in the process.
Documentation plays a key role in a QMS. Accurate records facilitate traceability and accountability. However, many organizations struggle with maintaining thorough documentation. This often leads to compliance issues and potential product failures. Consistent training on documentation practices can mitigate these challenges.
**Tip:** Implement a review system for documents to ensure they are up-to-date and accessible.
Investing in a strong QMS not only helps meet regulatory requirements but also fosters a culture of quality within the workforce. Quality should be everyone's responsibility, not just the quality assurance team.
| Quality Management Aspect | Description | Importance | Frequency of Review |
|---|---|---|---|
| Regulatory Compliance | Ensuring adherence to medical device regulations and standards. | Critical - ensures safety and legality of the product. | Annual |
| Risk Management | Identification and mitigation of potential risks associated with products. | High - minimizes harm to users and increases reliability. | Quarterly |
| Supplier Quality Management | Evaluation and monitoring of suppliers to ensure component quality. | Essential - influences overall product quality. | Semi-Annual |
| Training and Competence | Ongoing education and training for staff involved in production. | Very Important - affects production quality and compliance. | Bi-Annual |
| Product Testing | Rigorous testing of devices to ensure they meet performance criteria. | Vital - guarantees efficacy and safety before release. | Per Batch |
Conducting thorough risk assessments is pivotal in ensuring the safety of medical devices. Each device presents unique challenges, requiring a comprehensive understanding of its potential hazards. In the development stage, identify risks associated with design flaws, materials, and user errors. Engage experts from various fields to gather diverse insights. Their collective expertise can reveal unforeseen issues that may compromise safety.
Effective risk assessment involves continuous monitoring throughout the device lifecycle. Gathering data on device performance post-launch is essential. This phase often uncovers defects or safety concerns that were not apparent during initial testing. Regular audits and feedback from healthcare professionals help identify areas for improvement. However, organizations may face challenges with data collection and analysis. Inadequate tracking systems can hinder effective risk management.
Consider the human element in safety assessments. User behaviors and training can significantly impact device safety. Misunderstandings of instructions can lead to mishaps. Companies must refine their communication strategies to enhance understanding. Conducting iterative user-testing can shed light on issues in real-world settings. It ensures that devices not only function as intended but are also user-friendly and safe. Understanding and addressing these complexities can lead to better outcomes in medical device safety.
This chart illustrates the importance levels of various key factors that contribute to the quality and safety of medical devices. Conducting thorough risk assessments, maintaining strict quality control, adhering to regulatory compliance, considering user feedback, and validating designs are crucial aspects that should be emphasized in the medical device industry.
In the realm of medical devices, establishing robust testing and validation procedures is crucial. This ensures that the products meet stringent quality standards. The testing phase should involve real-life simulations. This helps identify potential failure points early in the process. Engineers need to collaborate closely with regulatory bodies. Their insights can guide the testing parameters and validation methods.
Quality assurance also demands continuous evaluation. After initial testing, teams must re-evaluate the devices as technologies evolve. This iterative process can reveal vulnerabilities. Stakeholders should regularly review feedback from clinical trials. Often, user experiences highlight overlooked issues. Addressing these concerns is essential for improving device reliability.
Documentation plays a vital role in this process. Clear records of testing results help establish accountability. They also serve as a reference for future projects. However, relying solely on past successes can be misleading. Each device has unique challenges. Embracing a culture of open feedback and reflection within teams can foster innovation. This adaptability is key to maintaining quality in an ever-changing field.
Medical devices must comply with strict regulatory requirements. This ensures they are safe and effective for users. Regulatory bodies outline detailed guidelines. These can often seem overwhelming, but they are crucial. Keeping up with these regulations is not just necessary; it is a responsibility. Every team member must understand their role in compliance.
Regular audits can help identify gaps. However, many organizations overlook the importance of consistent training. Staff need to be educated on compliance updates. Integrating compliance into daily operations fosters a culture of accountability. This approach has proven effective in other sectors. Yet, the medical device industry often struggles with this integration.
Documenting processes is equally essential. Accurate records help in tracing issues if they arise. Still, many companies fail to maintain thorough documentation. This can lead to serious repercussions. Regular review of compliance practices should become standard. It is an ongoing journey, not a one-time task. Embracing a proactive mindset can greatly improve outcomes.
: A robust QMS enhances product safety and efficacy. It ensures compliance with regulatory standards.
Regular audits and updates are essential. This keeps the system effective and aligned with industry needs.
Accurate records of all procedures are crucial. They facilitate traceability and accountability throughout the production.
Consistent training can help teams maintain thorough documentation. Regular reviews ensure that documents are up-to-date.
Testing ensures products meet quality standards. Real-life simulations help identify potential failure points early.
Clinical trial feedback often reveals overlooked issues. Addressing these concerns enhances overall device performance.
Many struggle to document processes thoroughly. This can lead to compliance issues and product failures.
Teams must continuously evaluate devices as technologies evolve. This helps identify new vulnerabilities and areas for improvement.
To ensure Key Factors in Medical Device Quality, it is essential to identify and adhere to essential quality standards specific to the industry. This involves the implementation of effective Quality Management Systems (QMS) throughout the production process, which helps in maintaining a consistent level of quality. Additionally, conducting thorough risk assessments is crucial to identify potential safety issues, thereby ensuring that devices are safe for use.
Robust testing and validation procedures play a significant role in the development of medical devices, ensuring that every product meets the necessary functional and safety requirements. Furthermore, maintaining compliance with regulatory requirements and guidelines is vital to uphold the integrity of the medical device. By focusing on these key aspects, manufacturers can achieve high-quality outcomes and ultimately enhance patient safety and satisfaction.

 MDV Medical
MDV Medical