✓
APPLICATION
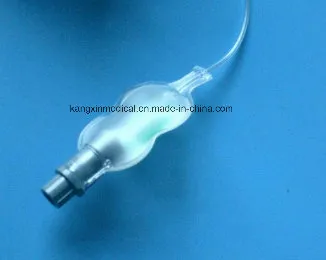
This is a compression device designed to assist hemostasis of the artery after a transradial procedure.
✓
STRUCTURE & FEATURE
The compression device is available in two specialized model types:
- Airbag Type: Composed of a compression airbag, an air-injection port, and a tie strap. It includes an appropriative inflator specifically for injecting air and adjusting inlet/outlet volume.
- Plate Type: Composed of a compression plate, bleeding-stopping pad, and an elastic band.
Note: This device is for single use only.