🌐 Cardiology Device Landscape in Uruguay
Uruguay has emerged as a sophisticated hub for medical technology in the Southern Cone. With a healthcare system that prioritizes universal coverage and advanced specialized care, the demand for high-quality Cardiology Devices has seen exponential growth. Manufacturers and factories catering to the Uruguayan market are not only focusing on local supply but are increasingly positioning themselves as strategic partners for the MERCOSUR region.
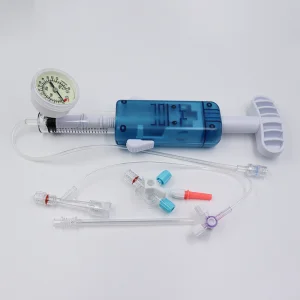
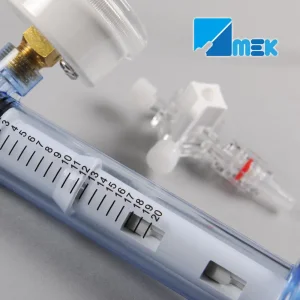
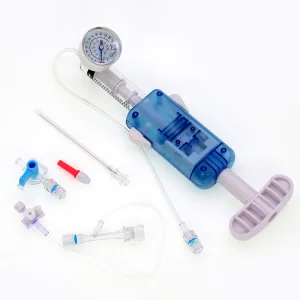
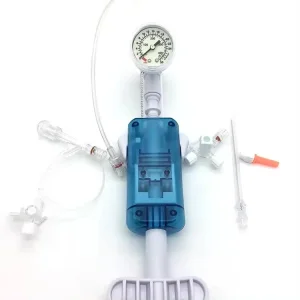
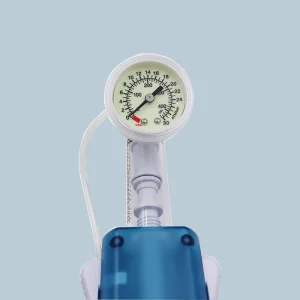
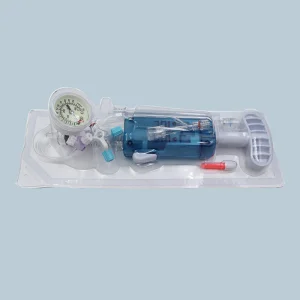
In the heart of Montevideo and the industrial zones of Canelones, the medical manufacturing sector is undergoing a digital transformation. The integration of AI-driven diagnostics and precision-engineered interventional tools, such as the PTCA Balloon Inflation Devices, reflects Uruguay’s commitment to adopting international standards like ISO 13485 and CE certifications. This ensures that every cardiac catheterization lab from the Hospital de Clínicas to private institutes in Punta del Este has access to world-class equipment.
Industry Trends: The Rise of Minimally Invasive Procedures
The global shift toward minimally invasive cardiovascular interventions is highly evident in Uruguay. Local healthcare providers are increasingly demanding disposable devices that offer higher pressure thresholds (up to 40 ATM) and ergonomic designs. This trend is driven by the need to reduce patient recovery time and hospital stays, a key metric for the Uruguayan Ministry of Public Health (MSP).
- Digital Integration: New cardiology devices are being designed to interface with hospital information systems (HIS) for real-time data logging during angioplasty.
- Sustainability: Factories are researching biodegradable components for disposable kits to align with Uruguay's strict environmental regulations.
- High-Pressure Precision: The move from 20ml to 25ml and 30ml inflation devices allows for more complex vessel dilation in multi-vessel coronary artery disease.
 MDV Medical
MDV Medical