
Optimized for quiet home environments in Seoul and metropolitan apartments.

High-efficiency sterilization compliant with Korean environmental regulations.

Advanced physical therapy tool used in top Incheon rehabilitation centers.

Portable and cost-effective solution for seasonal respiratory care.
South Korea has rapidly emerged as a global epicenter for medical device innovation, specifically within the respiratory equipment manufacturing sector. Driven by the "K-Medical" initiative, factories in Korea and their strategic partners are integrating high-speed internet (5G), Artificial Intelligence (AI), and advanced material sciences to redefine patient care. The landscape for respiratory equipment suppliers in Korea is no longer just about mechanical ventilation; it is about smart, interconnected ecosystems that support both hospital ICUs and the growing home-care demographic.
Major industrial hubs like Wonju Medical Device Techno Valley and clusters in Incheon Songdo have created a fertile ground for R&D. These regions facilitate a unique synergy between hardware engineers and software developers. Consequently, respiratory equipment factories are now producing devices that feature real-time data monitoring and AI-driven predictive analytics, which are essential for treating chronic obstructive pulmonary disease (COPD) and other long-term respiratory conditions prevalent in Korea’s aging population.
As South Korea faces a super-aging society, the demand for home-based medical devices has skyrocketed. Oxygen concentrators, portable nebulizers, and CPAP machines are now designed with a "lifestyle-first" approach—sleek, quiet, and user-friendly for the elderly. Suppliers are focusing on VPSA (Vacuum Pressure Swing Adsorption) technology to ensure high purity and durability, reflecting the local preference for "Premium Quality" (Pum-jil) in every electronic appliance.

Qingdao MDV Medical features a 50-person specialized team ensuring compliance with global and Korean medical standards.

Our production base includes a 1,500 sqm cleanroom and advanced quality inspection zones for high-precision equipment.

ISO 13485 and CE certified, meeting the rigorous regulatory requirements of the Korean Ministry of Food and Drug Safety (MFDS).
Qingdao MDV Medical Appliances Co., Ltd. is a modern enterprise focusing on the R&D, manufacturing and sales of anesthetic medical products, traditional Chinese medicine acupuncture, interventional products, injection puncture and nursing medical equipment. The company has passed the ISO 13485 Medical Device Quality System Certification, ensuring that every piece of respiratory equipment meets the highest international safety benchmarks.
Always taking the tenet of creating high-quality medical equipment, caring for life and health, and providing customers with first-class products and service, our company have been constantly upgrading our existing products and developing new products. We have many patented products to meet the requirements of product serialization and specialization, especially catering to the demanding needs of the Korean medical supply chain.
The industry is witnessing a shift toward precision respiratory medicine. Modern respiratory equipment suppliers in Korea are focusing on the integration of biocompatible materials. For instance, the transition from standard PVC to 100% Medical-Grade PVC and silicone materials is a response to the "Eco-Friendly Healthcare" trend popular in Seoul's top-tier university hospitals like SNUH and Asan Medical Center.
The core of this industry lies in the manufacturing machinery. Single-screw extrusion equipment for medical-grade catheters and nasal cannulas allows for high-precision manufacturing. This ensures that every tracheal intubation tube or infusion tube produced meets the sub-millimeter tolerances required for neonatal care and complex surgeries.
Furthermore, the focus on Face Mask Tightness Testing has become a cornerstone of the post-pandemic industry. In regions like Jinan and surrounding industrial zones, computer-controlled testing equipment is used to validate the efficacy of respiratory protective supplies, ensuring they meet the KMOEL (Korean Ministry of Employment and Labor) standards for occupational safety.

Interactive exhibit for health science museums and educational facilities.

Professional grade oxygen delivery for intensive care units.
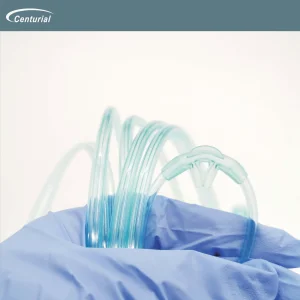
Biocompatible materials for safe, long-term pulmonary use.

Robust protection for emergency responders and industrial safety.
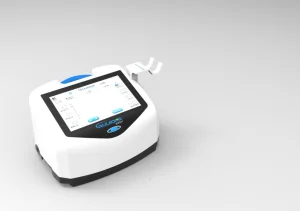
Effective airway clearance technology for chronic lung disease patients.

Tailored breathing solutions for the growing veterinary sector in Korea.
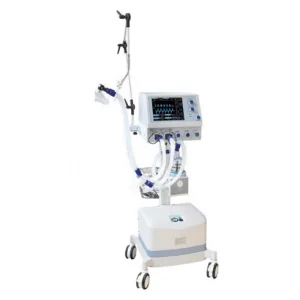
Hospital-grade breathing apparatus with multiple ventilation modes.

Durable air supply systems for high-risk industrial environments.



Always taking the tenet of creating high-quality medical equipment, caring for life and health, and providing customers with first-class products and service, our company have been constantly upgrading our existing products and developing new products. From the precision required in Medical-Grade Single-Screw Extrusion for nasal cannulas to the real-time monitoring of Etco2 in patient monitors, our mission is to provide the Korean medical community with reliable, innovative tools.
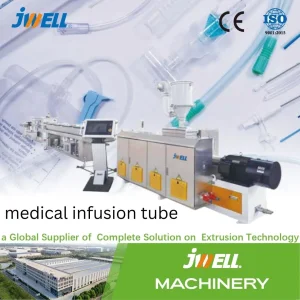
For high-precision Tracheal Intubation and Nasal Cannula manufacturing.

Essential for ICU respiratory care and patient safety tracking.

Ensuring the highest safety standards for respiratory protective supplies.
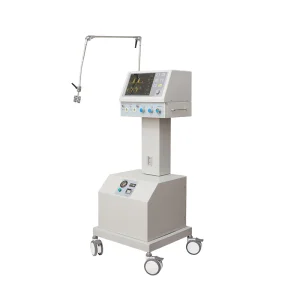
Advanced support for clinical hospitals and emergency centers.
Choosing the right respiratory equipment factory requires a balance of innovative technology and reliable manufacturing standards. For the Korean market, where quality is non-negotiable, our products offer:
Whether you are a hospital administrator in Seoul looking for the next generation of ICU ventilators, or a medical supplier in Gyeonggi-do seeking reliable oxygen concentrators, our team is ready to deliver world-class solutions.