
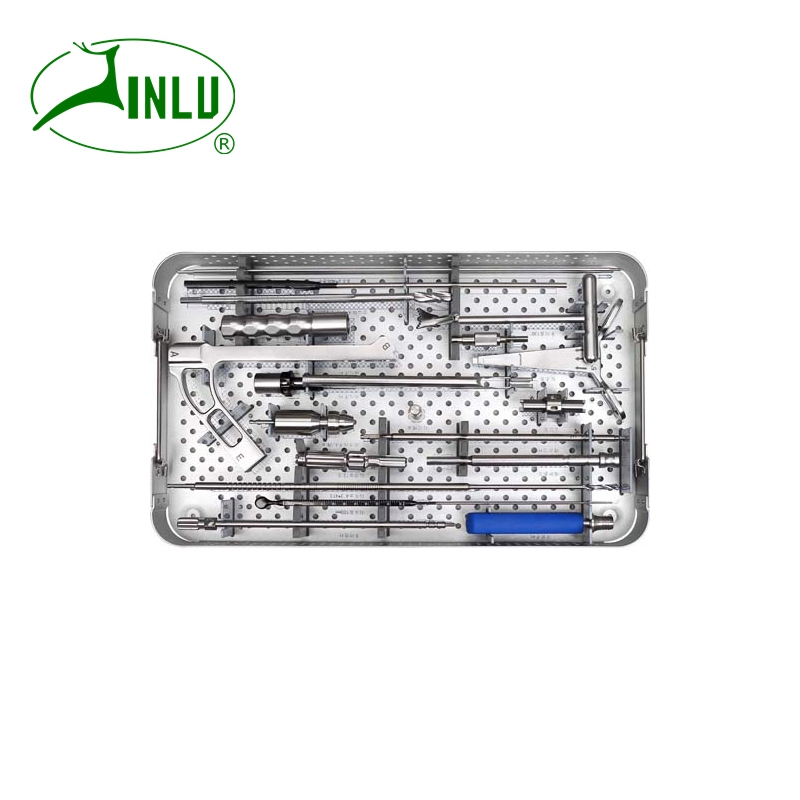










| Femoral Neck System Instrument Set | |||
|---|---|---|---|
| P/N | NO. | Description | Size |
| 37101 | 1 | Guider | 130° |
| 37102 | 2 | Calibrating Instrument | - |
| 37103 | 3 | Threaded Guide Pin | φ3.2*400 |
| 37104 | 4 | Guide Pin Depth Cauge | - |
| 37105 | 5 | cannulated drill | φ10.2 |
| 37106 | 6 | reamer | φ12.5 |
| 37107 | 7 | reamer nut | - |
| 37108 | 8 | impactor handle 1 | - |
| 37109 | 9 | impactor handle 2 | - |
| 37110 | 10 | multifunction rod | - |
| 37111 | 11 | drill bit | φ4.3 |
| 37112 | 12 | drill stop | - |
| 37113 | 13 | protection sleeve | - |
| 37114 | 14 | quick coupling torx head screw driver | T25 |
| 37115 | 15 | quick coupling hex screw driver | SW3.5 |
| 37116 | 16 | cannular hammer | - |
| 37117 | 17 | depth cauge | 100mm |
| 37118 | 18 | quick coupling torque handle | 4.0N.m |
| 37119 | 19 | straight quick coupling handle | - |
| 37120 | 20 | connection for quick coupling handle | - |





Established in 1958, we are engaged in the development, manufacture, and sales of orthopedic implants and instruments. We are recognized as a key enterprise for medical apparatus and hold a director position within the Medical Apparatus Industry Association. The product line covers spine pedical screws, interlocking nails, locking plates/screws, trauma plates/screws, cannulated screws, external fixators, hip prosthesis, and surgical instruments.
Our products are characterized by a wide variety and complete specifications. All medical apparatus have passed approval and authentication from national and provincial offices. Our advanced manufacturing equipment includes imported CNC, high-precision digital control machines, and a 100,000 Grade axenic purification chamber to ensure high-quality production.
