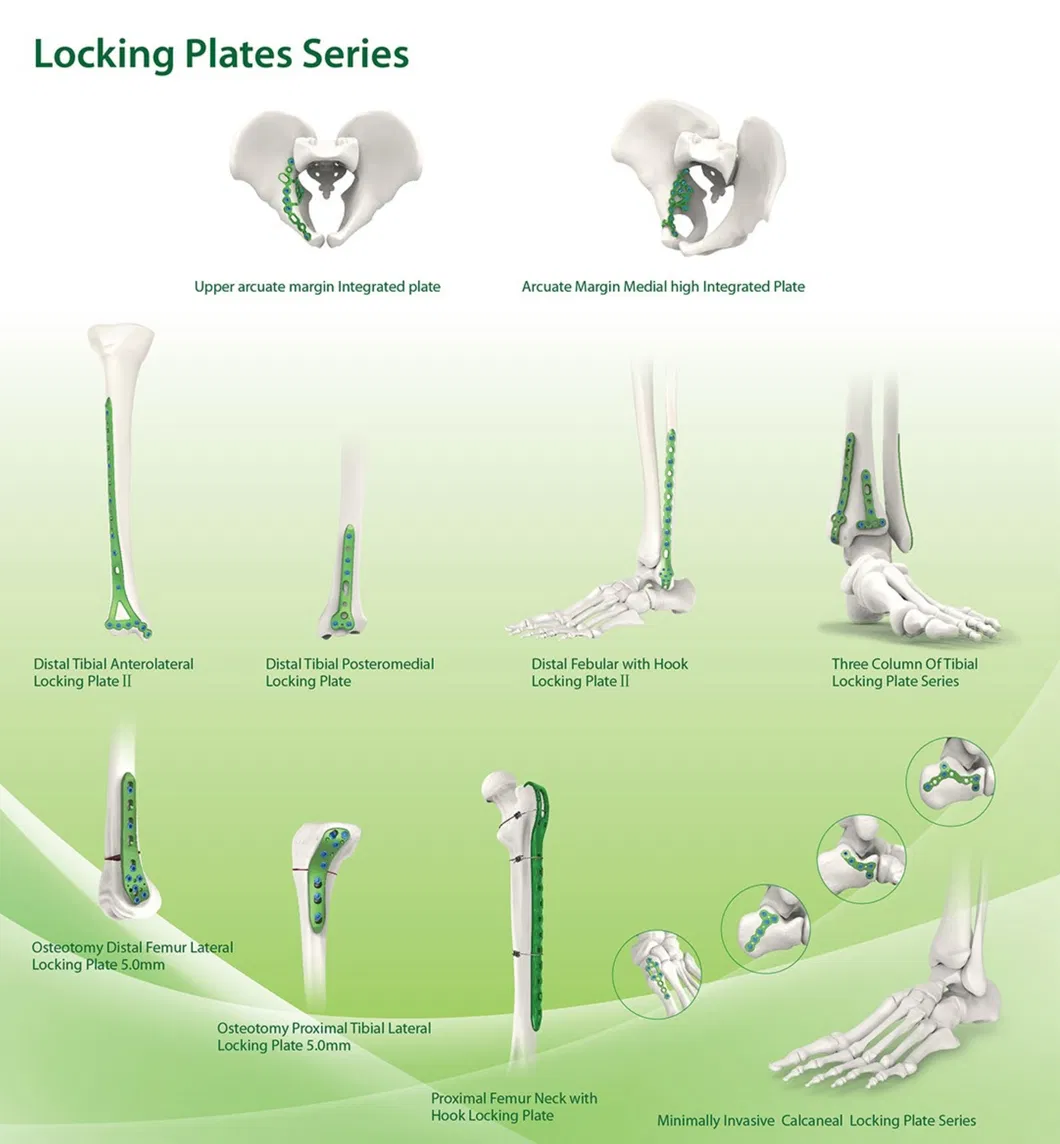
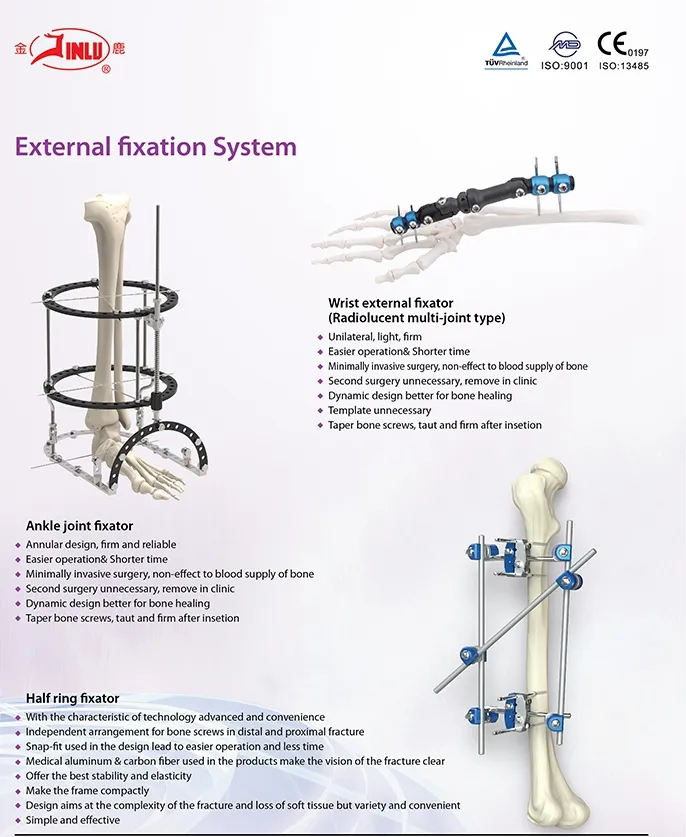
What are your primary orthopedic product lines?
We specialize in metal bone plate screw systems, cannulated bore screws, interlocking intramedullary nails, spinal fixation devices, and external fixation systems.
How do you ensure the quality of your implants?
All products are produced according to EU CE standards. We conduct pre-production sample analysis and a 100% final inspection before shipment to ensure safety and precision.
Which materials are used for your cage fusion devices?
We primarily use high-grade Titanium and medical-grade PEEK material, both known for their excellent biocompatibility and mechanical strength in spinal procedures.
What certifications does the manufacturing facility hold?
Our facility is ISO 9001 and ISO 13485 certified, and our products carry CE and FDA marks, adhering to global medical device regulations.
What is your monthly production capacity?
We have a stable production capacity of approximately 10,000 pieces per month, supported by advanced CNC and high-precision digital milling machines.
Do you provide international shipping for orders?
Yes, we support various delivery terms including FOB, CIF, and EXW, and ship globally via Express Delivery (DHL/TNT/UPS/FedEx) or Air Freight.