











Advanced Cervical Peek Cage Fusion Device and Spine Orthopedic Implants designed for superior stability and surgical precision.

| P/N | Product Name | Size | Unit |
|---|---|---|---|
| 1334-T001 | Telescopic body prosthesis | 12*20mm | pcs |
| 1334-T002 | 12*28mm | pcs | |
| 1334-T003 | 12*35mm | pcs | |
| 1335-T001 | Cervical cage (PEEK) | 6*16*13 | pcs |
| 1335-T002 | 7*16*13 | pcs | |
| 1335-T003 | 8*16*13 | pcs | |
| 1335-T004 | Lumbar Cage (PEEK) | 8*22mm | pcs |
| 1335-T005 | 8*26mm | pcs | |
| 1335-T006 | 10*20mm | pcs |
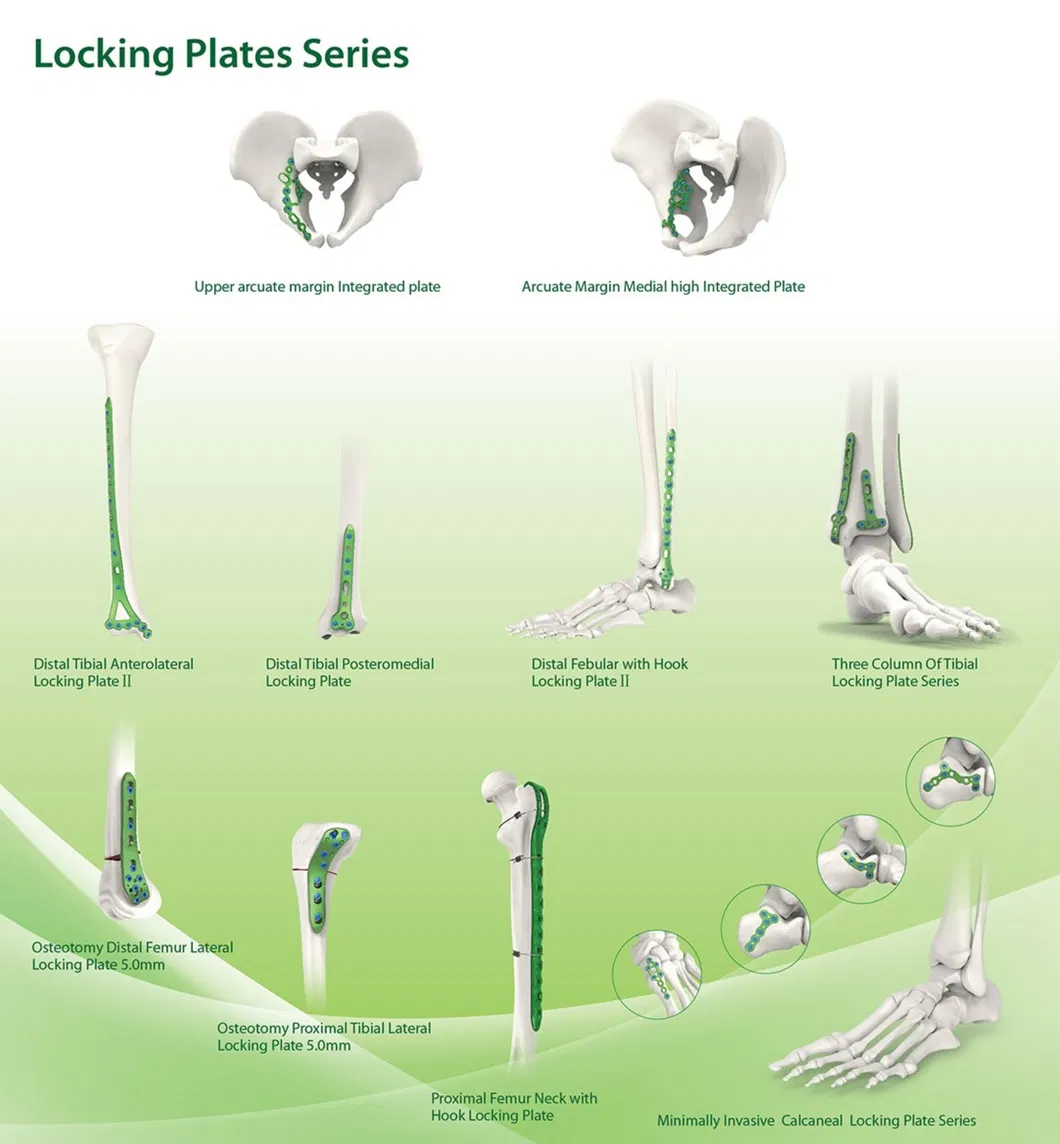
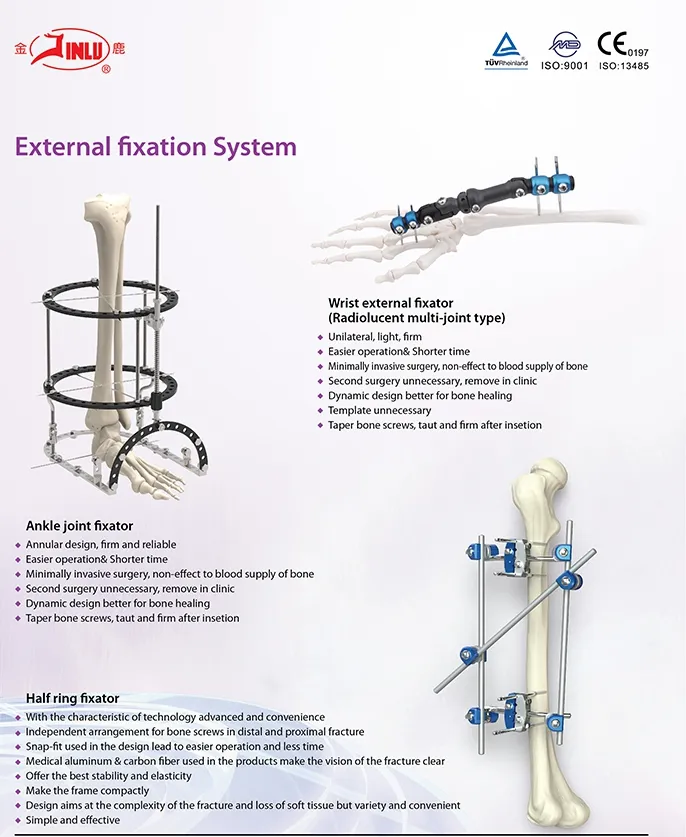






Established in 1958, our facility is dedicated to the development, manufacture, and sales of orthopedic implants and instruments. We are recognized as a key enterprise in the national medical apparatus industry. Our comprehensive product line covers spine pedicle screws, interlocking nails, locking plates, trauma screws, cannulated screws, external fixators, hip prosthesis, and specialized surgical instruments.
Our manufacturing process utilizes advanced equipment including imported CNC centers, high-precision digital control machines, and a 100,000 Grade axenic purification chamber to ensure the highest quality standards. We adhere to the tenet of "creating high quality with science and technology" and provide innovative solutions for orthopedic surgery worldwide.
