



1 / 5











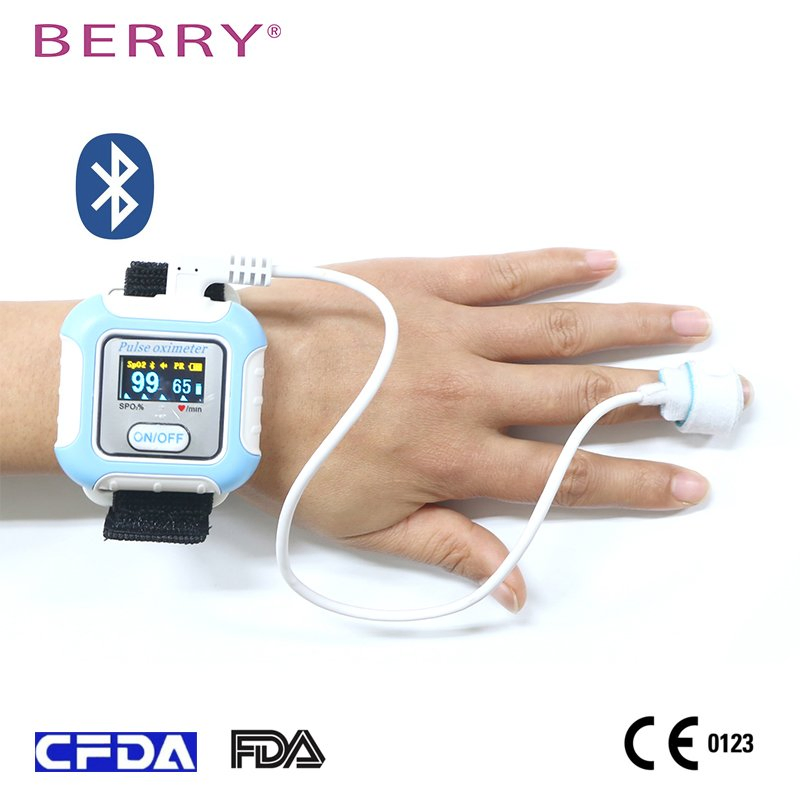
Suitable for sleep hypopnea syndrome monitoring. This Sleep Apnea Screening Monitor holds 510K, CE0123, ISO13485, FCC, and RoHS certifications. It accurately captures pulse signals, as validated in numerous clinical trials, meeting high standards for blood oxygen and pulse rate measurements.

BM2000A is applicable for Aviation, Snorers, Children, Asthma, and COPD patients.
Download the SmartHealth APP.
Wear equipment for at least 6 hours.
Apply foam SpO2 sensor to fingertip.
Transfer data via Bluetooth.
View and share the reports.
| Parameter | Specification |
|---|---|
| Display | SpO2, PR |
| Battery | 3.7V (1000mAh) Rechargeable Lithium |
| Pulse Rate Range | 25 - 250bpm (Accuracy: ±2bpm) |
| SpO2 Range | 35% - 100% (Accuracy: ±2% for 80-100%) |
| Dimensions | 68(L) x 58(W) x 20(D) mm |
| Weight | 50g |
| Working Voltage | DC 3.4V ~ DC 4.3V |
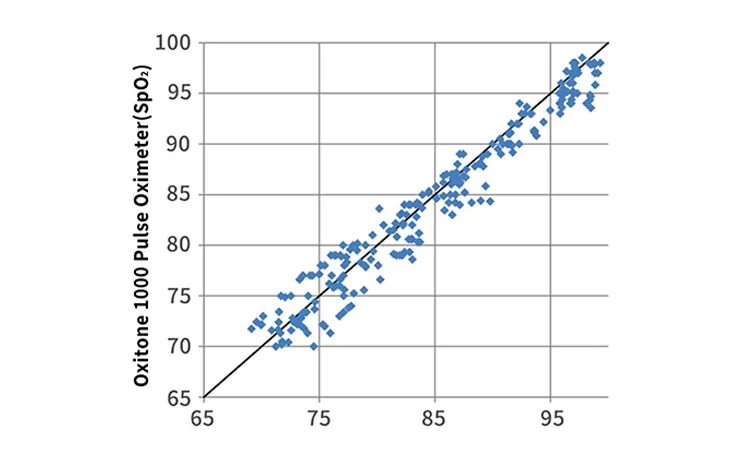
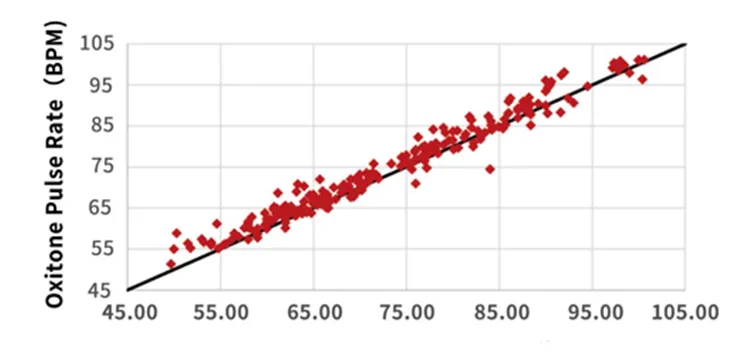
Correlation = 0.98, P-value < 0.0001 vs Reference ECG
| Comparison: Reference Pulse Oximeter | |||||
|---|---|---|---|---|---|
| Pulse Oximeter Range | 70 ~ 100 | 90 ~ 100 | 80 - < 90 | 67 - < 80 | Result |
| Error | -0.6 | -1.1 | -0.9 | 0.1 | PASS |
| RMS Error | 1.9 | 1.7 | 2.0 | 2.1 | |
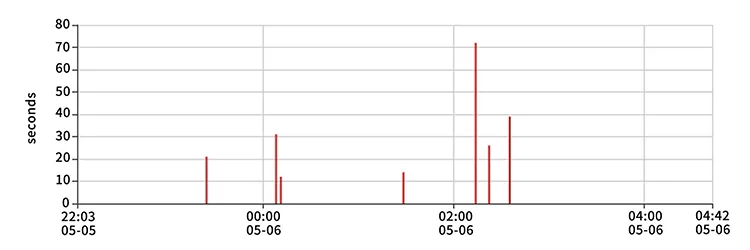
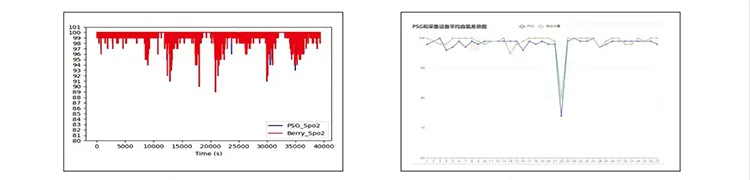
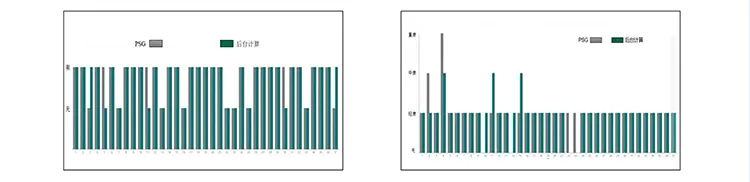
| Analysis Of Clinical Trial Results | ||
|---|---|---|
| Average Accuracy | 98.51% | |
| Hypoxemia Diagnosis | Sensitivity: 89.29% | Accuracy: 92.59% |
| OSAHS Judgment | Sensitivity: 94.17% | Accuracy: 89.62% |

