




1 / 5










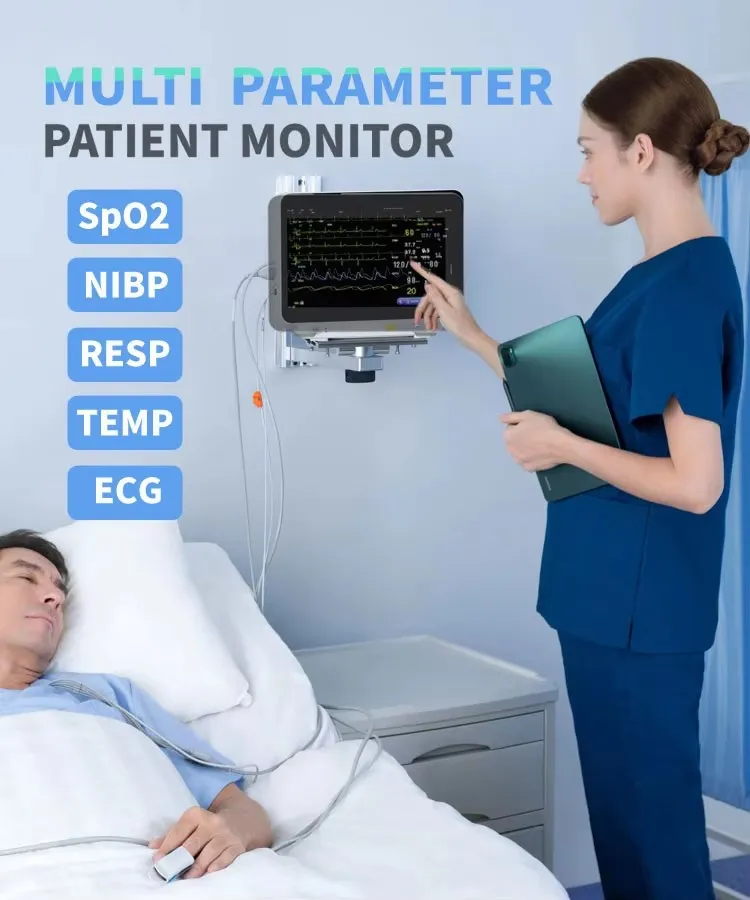

| Display | 7-inch color TFT LCD |
| Electrical Specifications | 12V 2A Adapter |
| Battery | Rechargeable lithium battery, both AC and DC supported |
| Working Method | Continuous work |
| Central Monitoring System | Support |








